A prep is considered successful if it is good or excellent 2,3
- 97% agreement was seen between central and local readers3
SUTAB® Split-Dose Regimen†
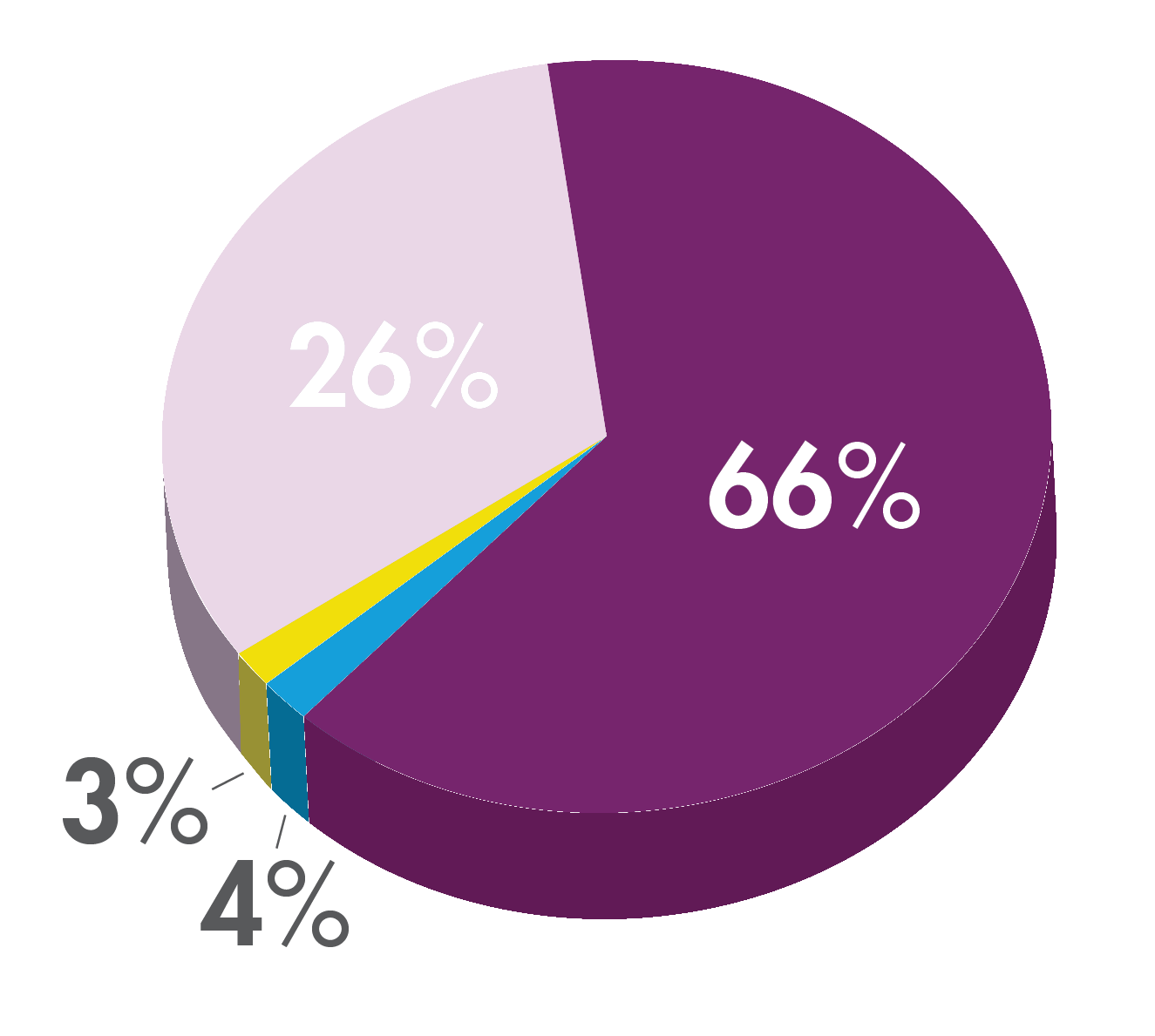
A prep is considered successful if it is good or excellent 2,3
SUTAB® Split-Dose Regimen†
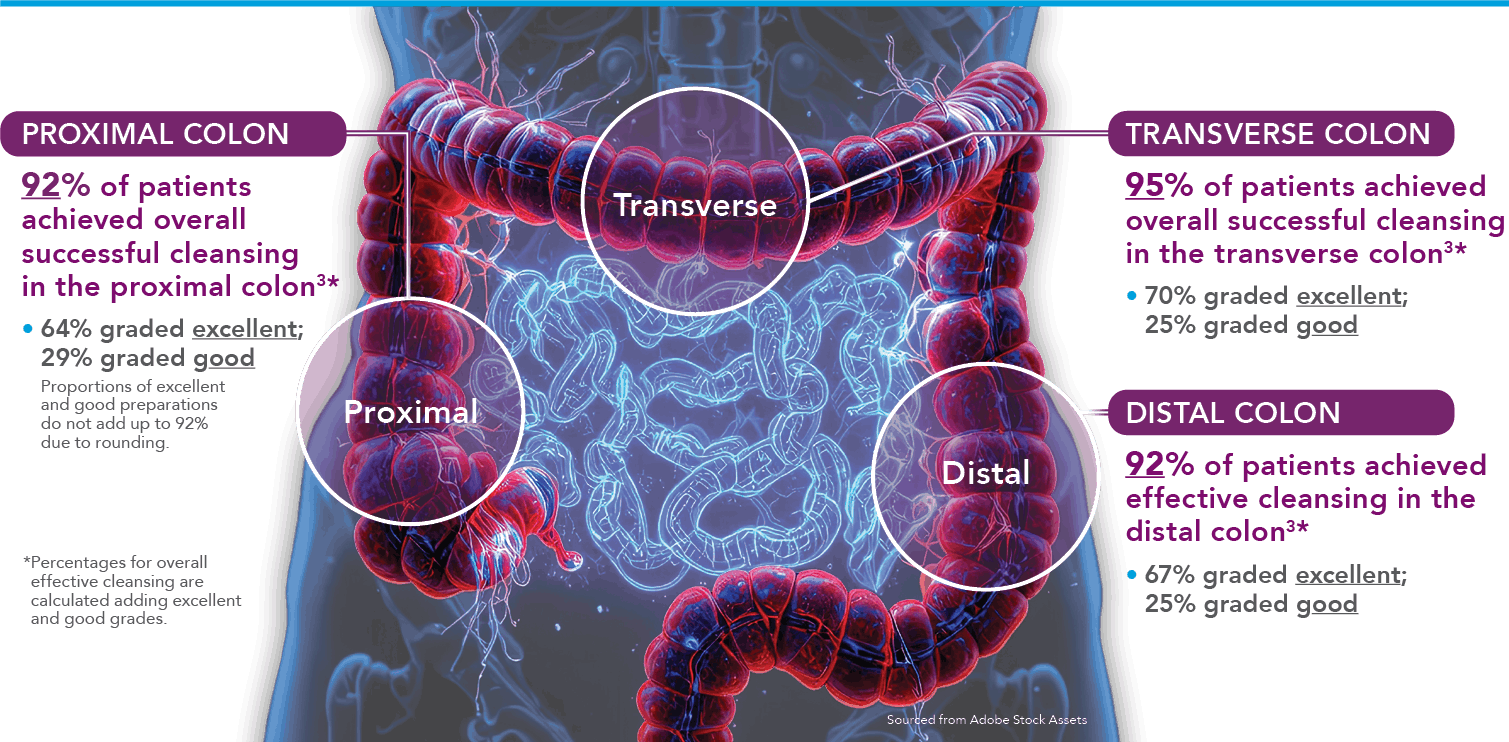
Study Design3: Study 1 was a phase 3 trial that was conducted in 22 US sites. Of the 620 subjects who enrolled, 548 patients were evaluable for efficacy analyses. The primary endpoint was the percentage of subjects with successful cleansing, which was based on the US FDA Bowel Prep Scoring Scale, the goal of which was to determine non-inferiority against MoviPrep®. Safety follow-up visits were required 24-48 hours after colonoscopy and on days 7 and 30 if adverse events or lab abnormalities were indicated. A subset of colonoscopies was graded by central readers (exploratory). The primary endpoint of non-inferiority was met.
| Hard-to-Prep Patients (n=115) | Successful Cleansing Rate |
Excellent Cleansing Rate |
|---|---|---|
|
History of constipation Opioid use Failed colonoscopy BMI ≥35 |
90% | 61% |
| Afternoon colonoscopy (n=49) | 96% | 74% |
| Endpoint | SUTAB Result |
|---|---|
| Cecal intubation rate | 98% |
| Preparation adequacy | 97% |
| Procedure duration (median) | 13.5 minutes |
| Intraprocedural water volume (median) | 30 mL |
Recommended Cecal Intubation Rate: ≥95%
ACG®=American College of Gastroenterology
ASGE®=American Society for Gastrointestinal Endoscopy
SUTAB® (sodium sulfate, magnesium sulfate, and potassium chloride) tablets, for oral use
Indications and Usage
SUTAB is indicated for the cleansing of the colon as a preparation for colonoscopy in adults.
ADDITIONAL IMPORTANT SAFETY INFORMATION
Contraindications
SUTAB is contraindicated in the following conditions:
Warnings and Precautions
Adverse Reactions
Most common gastrointestinal adverse reactions are nausea, abdominal distension, vomiting, and upper abdominal pain.
These are not all the possible side effects of SUTAB. Please see Adverse Reactions (6) in the Prescribing Information for a full list.
Drug Interactions
Drugs that increase risk of fluid and electrolyte imbalance. (7.1)
To Report SUSPECTED ADVERSE REACTIONS, contact Azurity Pharmaceuticals, Inc. at 1-800-461-7449, or FDA at 1-800-FDA-1088 or www.fda.gov/MedWatch.
SUTAB® is a registered trademark of Azurity Pharmaceuticals, Inc.
PP-STB-6510-US-EN-v1
View the Full Prescribing Information and Medication Guide.
References: 1. IQVIA, National Prescription Audit Report. 2. SUTAB® [package insert]. Woburn, MA: Azurity Pharmaceuticals, Inc. 3. Di Palma JA, Bhandari R, Cleveland M, et al. A safety and efficacy comparison of a new sulfate-based tablet bowel preparation versus a PEG and ascorbate comparator in adult subjects undergoing colonoscopy. Am J Gastroenterol. 2021;116(2):319-328. doi: 10.14309/ajg.0000000000001020. 4. Rex DK, Johnson DA, Anderson JC, et al; American College of Gastroenterology. American College of Gastroenterology guidelines for colorectal cancer screening 2009 [corrected]. Am J Gastroenterol. 2009;104(3):739-750. 5. Rex DK, Boland CR, Dominitz JA, et al. Colorectal cancer screening: recommendations for physicians and patients from the US Multi-Society Task Force on Colorectal Cancer. Am J Gastroenterol. 2017;112(7):1016-1030. doi:10.1038/ajg.2017.174. 6. Rex DK, Anderson JC, Butterfly LF, et al. Quality indicators for colonoscopy. Gastrointest Endosc. 2024;100(3):352-381. doi:10.1016/j.gie.2024.04.2905 7. Data on file. Woburn, MA: Azurity Pharmaceuticals, Inc. 8. SUFLAVE® [package insert]. Woburn, MA: Azurity Pharmaceuticals, Inc. 9. SUPREP® Bowel Prep Kit [package insert]. Woburn, MA: Azurity Pharmaceuticals, Inc. 10. Plenvu® [package insert]. Bridgewater, NJ: Bausch Health LLC. 11. Clenpiq® [package insert]. Parsippany, NJ: Ferring Pharmaceuticals, Inc. 12. Rex DK, Di Palma JA, Rodriguez R, et al. A randomized clinical study comparing reduced-volume oral sulfate solution with standard 4-liter sulfate-free electrolyte lavage solution as preparation for colonoscopy. Gastrointest Endosc. 2010;72(2):328-336. 13. Matro R, Daskalakis C, Negoianu D, et al. Randomised clinical trial: polyethylene glycol 3350 with sports drink vs polyethylene glycol with electrolyte solution as purgatives for colonoscopy—the incidence of hyponatraemia. Aliment Pharmacol Ther. 2014;40(6):610-619.
SUFLAVE® (polyethylene glycol 3350, sodium sulfate, potassium chloride, magnesium sulfate, and sodium chloride for oral solution)
Indications and Usage
SUFLAVE is indicated for the cleansing of the colon as a preparation for colonoscopy in adults.
ADDITIONAL IMPORTANT SAFETY INFORMATION
Contraindications
SUFLAVE is contraindicated in the following conditions:
Warnings and Precautions
Adverse Reactions
Most common adverse reactions (≥2%) are: nausea, abdominal distension, vomiting, abdominal pain and headache.
These are not all the possible side effects of SUFLAVE. Please see Adverse Reactions (6) in the Prescribing Information for a full list.
Drug Interactions
Drugs that increase risk of fluid and electrolyte imbalance. (7.1)
To Report SUSPECTED ADVERSE REACTIONS, contact Azurity Pharmaceuticals, Inc. at 1-800-461-7449, or FDA at 1-800-FDA-1088 or www.fda.gov/MedWatch.
SUFLAVE® is a registered trademark of Azurity Pharmaceuticals, Inc.
PP-SUF-6488-US-EN-v1